An MRI image of the brain shows the area of an Alzheimer’s patient.
Getty Images
The Food and Drug Administration on Friday granted fast-track approval for the Alzheimer’s drug lecanemab, the second treatment of Biogen and Japanese partner Eisai to receive the initial green light in less than two years.
The FDA approval comes after results of a clinical trial published in November showed that lecanemab slightly reduced cognitive decline in people with mild impairment from Alzheimer’s disease, but the treatment also carried the risk of brain swelling and bleeding.
Eisai, which leads the development of lecanemab, prices the treatment at $ 26,500 per year in the U.S. It will be sold under the name Leqembi.
The FDA can expedite approval of a drug to bring it to market quickly if it is expected to help patients with more serious illnesses than what is currently available. Biogen and Eisai applied for an accelerated approval in July.
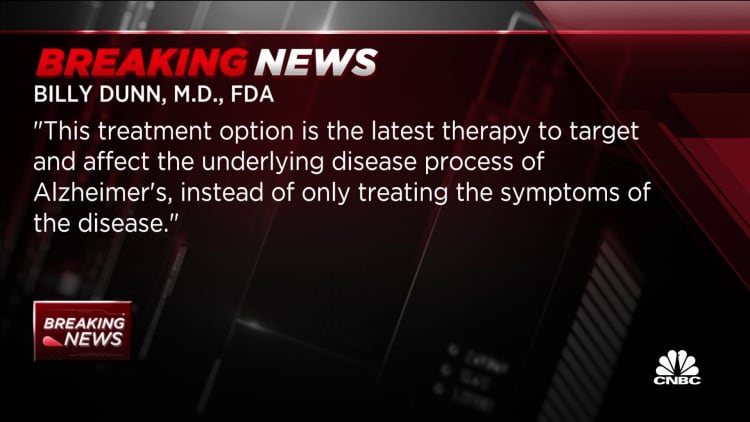
“Alzheimer’s disease immeasurably incapacitates the lives of those who suffer from it and has a devastating effect on their loved ones,” said Dr. Billy Dunn, director of the FDA’s neuroscience division, in a statement. “This treatment option is the latest therapy to target and influence the Alzheimer’s disease process, rather than just treating the symptoms of the disease.”
More than 6.5 million people in the US have Alzheimer’s disease. Irreversible diseases impair memory, thinking ability, and eventually the ability to perform simple tasks.
The decision on lecanameb comes after Congress issued a scathing report last week on how the FDA handled the controversial approval of another Alzheimer’s drug developed by Biogen and Eisai, called Aduhelm. The 2021 approval for the treatment, which experts say shows no clear clinical benefit, is “full of irregularities,” according to the report.
The congressional report said “FDA must take immediate action to ensure that the process of examining the treatment of Alzheimer’s disease in the future does not lead to the same doubts about the integrity of the FDA review.”
Modestly slows the disease
Lecanemab is a monoclonal antibody that targets a protein called amyloid that forms in the brain in people with Alzheimer’s. The antibody is given intravenously every two weeks in a dose determined by the patient’s body weight with 10 milligrams given per kilogram.
Lecanemab was approved by the FDA based on the reduction of amyloid plaques observed in clinical trial participants who received the treatment, according to a statement from the agency. Participants who received no treatment, the placebo arm, had no reduction in amyloid plaques.
The results of the clinical trial, published in the New England Journal of Medicine, found that cognitive decline was 27% slower over 18 months in those who received lecanemab compared to those who did not receive the treatment. The research was funded by Biogen and Eisai.
Cognitive decline is measured using a system called the clinical dementia rating, which is an 18-point scale with higher scores indicating greater levels of impairment. It measures cognitive functions such as memory, judgment and problem solving.
Alzheimer’s disease progressed by an average of 1.21 points in the group that received lecanemab compared to 1.66 points in the group that did not receive treatment, a small difference of 0.45 points.
Nearly 1,800 people aged 50 to 90 with early Alzheimer’s participated in the trial, about half of whom received lecanemab and half who did not.
Safety concerns
Although lecanemab can reduce cognitive decline, the treatment also has risks.
Nearly 13% of people who received lecanemab developed brain swelling compared with about 2% in the group that did not receive the treatment. However, most cases are mild to moderate, cause no symptoms, and usually resolve within four months.
About 3% of patients receiving lecanemab experienced more serious brain swelling with symptoms including headache, visual disturbances and confusion.
About 17% of people who received lecanemab had a brain bleed, compared with 9% in the group that did not take the treatment. The most common symptom associated with bleeding is dizziness.
Overall, 14% of people who received lecanemab experienced serious side effects in the clinical trial, compared to 11% of people who did not receive the treatment.
The study’s authors say longer clinical trials are needed to determine the efficacy and safety of lecanemab in patients with early Alzheimer’s disease.
The FDA said the prescribing information for lecanemab will include a warning about the risk of swelling and bleeding, commonly referred to as an amyloid imaging disorder.
The death of a clinical trial participant in the Chicago area may be related to lecanemab, according to a research letter published in the New England Journal of Medicine this week.
A 65-year-old suffered a stroke and was hospitalized four days after his third lecanemab infusion. A CT scan performed after the patient’s stroke revealed extensive bleeding in the brain. An MRI done 81 days before the stroke found no bleeding.
Patients also receive a drug, called t-PA, which is used to break up blood clots that cause strokes. But massive brain bleeding would be an unusual complication of this drug, according to the doctors who wrote the research paper.
Researchers involved in the clinical trial of lecanemab, in a response letter, stated that the blood clotting drug appeared to be the direct cause of the patient’s death, with the first symptoms occurring 8 minutes after receiving an infusion of the blood clot buster.